Illinois residents may purchase up to 30 grams of flower/pre-rolls; up to 500 milligrams of THC-infused edibles; and up to 5 grams of concentrates. Non-residents may purchase up to 15 grams of flower/pre-rolls; up to 2.5 grams of concentrates; and up to 250 milligrams of THC-infused edibles.


Order Now [IL, MD]
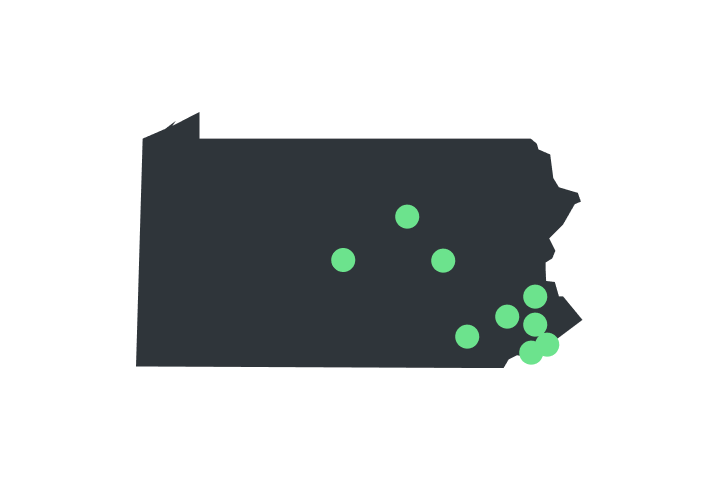
Order Now [PA]
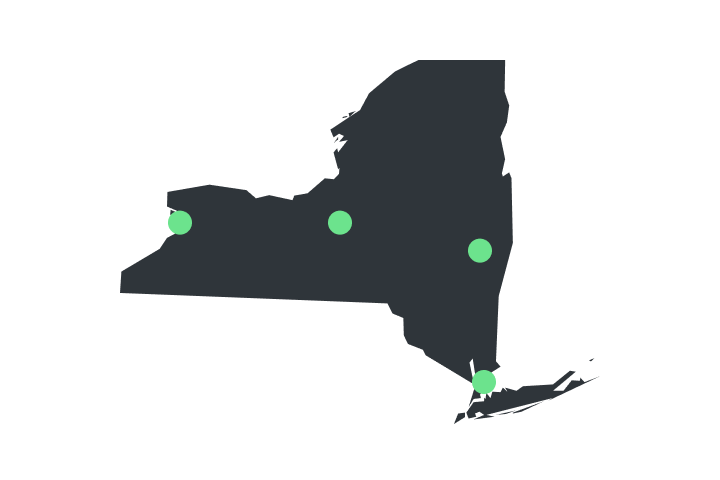
Order Now [NY]
Order Now – Shrewsbury
Order Now – Wareham
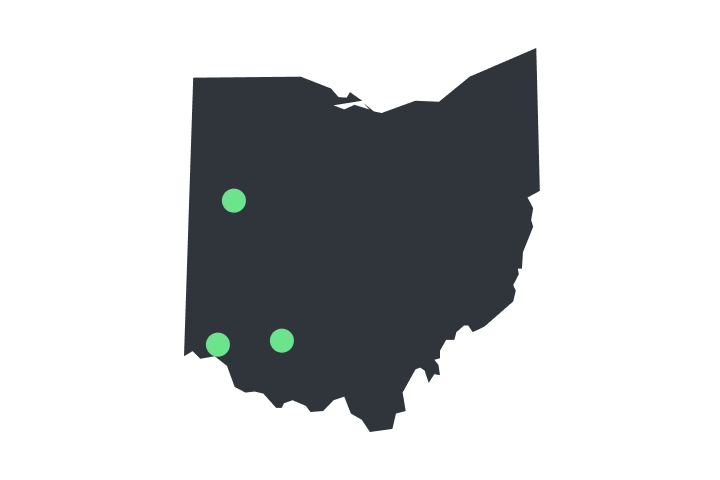
Order Now [OH]




Find a Dispensary Near You
Verilife dispensaries are located throughout Illinois, Maryland, Massachusetts, New York, Ohio, and Pennsylvania. Find your closest dispensary and start shopping today.


Fresh Picks
What's new and noteworthy at Verilife dispensaries.
Our Brands










IMAGE
IMAGE


Dispensary Discounts
Medical cannabis patients and adult-use customers save more with our ongoing offers.


Recreational Cannabis
Explore the adult-use program, including who is qualified to visit a dispensary, and how much you can purchase.
Medical Cannabis
Learn more about the Medical Cannabis Registry Program, including qualifications, purchase limits, and more.


Get a Medical Card
Check out our guide to getting a medical cannabis card, including a step-by-step application guide.


What are Cannabinoids & How Do They Work?
There are dozens of cannabinoids found in the cannabis plant, in addition to THC and CBD. Learn more about them in our guide.


What is the Endocannabinoid System?
You have a nervous system, immune system, and even a musculoskeletal system, but did you know you also have an endocannabinoid system?


Talk About Terpenes
Cannabis is known for its unique smells and flavors, which can be attributed to tiny compounds known as terpenes.